|
the ion with 1 more oxygen than the most common ionic form is called per.the ion with 2 fewer oxygens than the most common ionic form is called hypo- .the ion with 1 less oxygen than the most common ionic form is called -ite ion.The most common form of the oxoanions in a series is named by changing the ending of the central element's name to -ate and adding the word ion 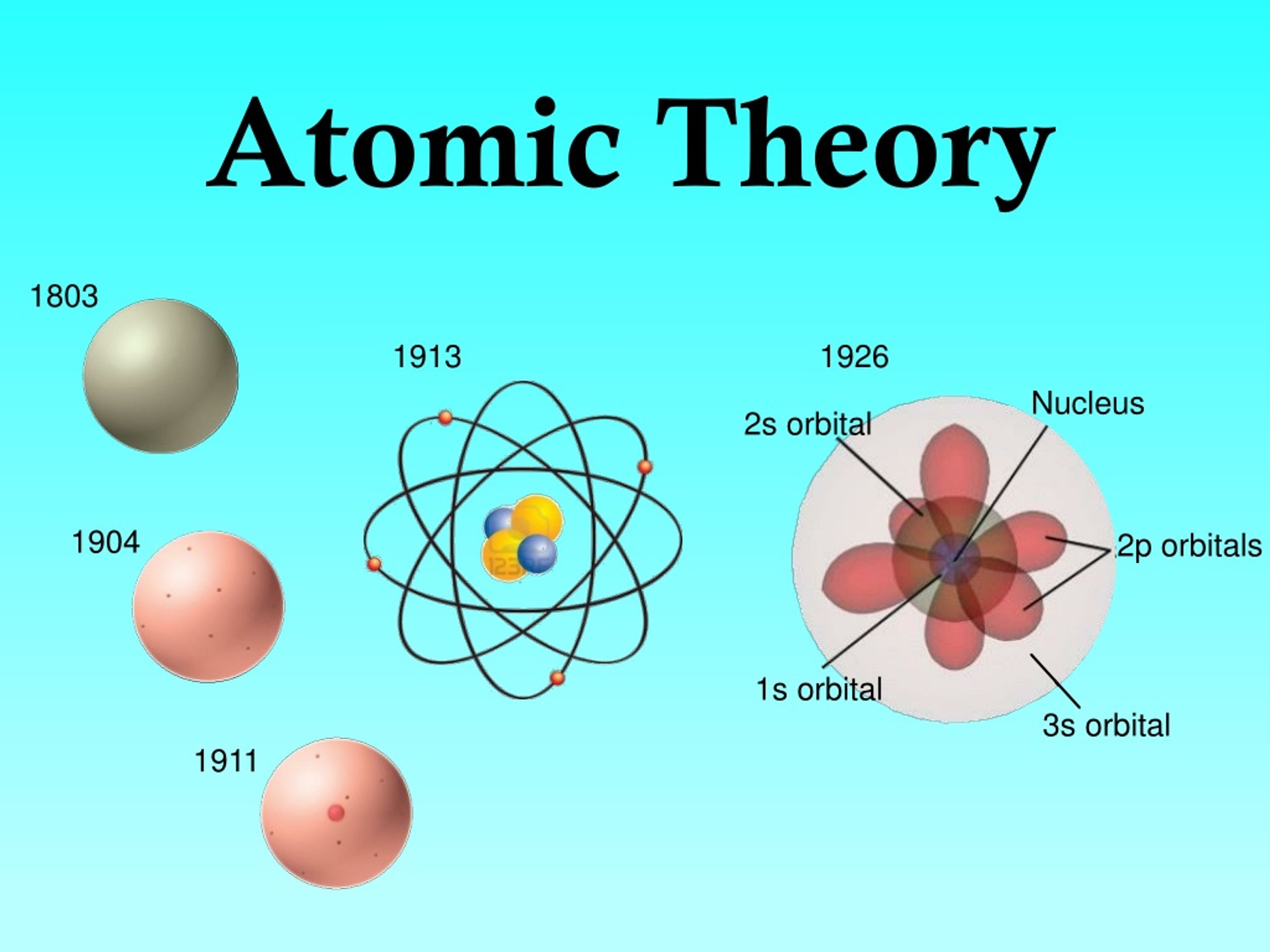 
They often occur in series of 2 or more ions with the same central atom but different numbers of oxygens Define isotopes and give examples for several elements. Describe the three subatomic particles that compose atoms. A melted wristwatch that survived the atomic bombing of Hiroshima on Aug. Summarize and interpret the results of the experiments of Thomson, Millikan, and Rutherford. Polyatomic anions that contain one or more oxygen atoms and one central atom of another element. By the end of this section, you will be able to: Outline milestones in the development of modern atomic theory. named by changing the ending of the element's name to -ide and adding the word ion.if there are more than one possible charge (especially for the transition metals), a Roman numeral is added behind the name of the elements to indicate the charge.named by adding the word ion to the name of the elements.Chemical reactions largely involve atoms or groups of atoms and the interactions between their electrons. In general, electrons are easier to add or remove from an atom than a proton or neutron. The modern atomic theory, proposed about 1803 by the English chemist John Dalton (Figure 2.3.1 2.3. The particles within an atom are bound together by powerful forces.Isotopes and ions of an atom with a constant number of protons are all variations of a single element. Varying the number of electrons results in ions. Varying the number of neutrons results in isotopes. The number of protons (also known as its atomic number) determines the element.Almost all of the mass of an atom is in its nucleus almost all of the volume of an atom is occupied by electrons.The average size of an atom is about 100 picometers or one ten-billionth of a meter. atomic theory, ancient philosophical speculation that all things can be accounted for by innumerable combinations of hard, small, indivisible particles (called atoms) of various sizes but of the same basic material or the modern scientific theory of matter according to which the chemical elements that combine to form the great variety of substa. Electrons can also move between orbitals. Technically, an electron can be found anywhere within the atom, but spends most of its time in the region described by an orbital. Some electron shells resemble spheres, but others look more like dumb bells or other shapes. Simple models show electrons orbiting the nuclear in a near-circular orbit, like planets orbiting a star, but real behavior is much more complex. Many consider 2008 the 200th anniversary of atomic theory, John Dalton’s momentous theory of the nature of matter. Electrons are organized into shells, which is a region where an electron is most likely found. Electrons move around outside the nucleus.The nucleus carries a positive electrical charge. The nucleus of an atom contains protons and neutrons. 
The mass of a proton is 1840 times greater than the mass of an electron. The mass of a proton is essentially the same as that of a neutron. Protons and neutrons are about the same size as each other and are much larger than electrons. 
As with any scientific theory, an atomic theory is based on scientific evidence available at any given time and serves to suggest future lines of research about atoms. In other words, neutrons do not have a charge and are not electrically attracted to either electrons or protons. Atomic theory 1 An atomic theory is a model developed to explain the properties and behaviors of atoms. Electrons and protons are electrically attracted to each other. Like charges (protons and protons, electrons and electrons) repel each other. The charge of a proton and an electron are equal in magnitude, yet opposite in sign.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
